Let us now look at the drawbacks of this theory. However, this theory is not entirely free of limitations. Learn about Atomic Mass and Molecular Mass here. 
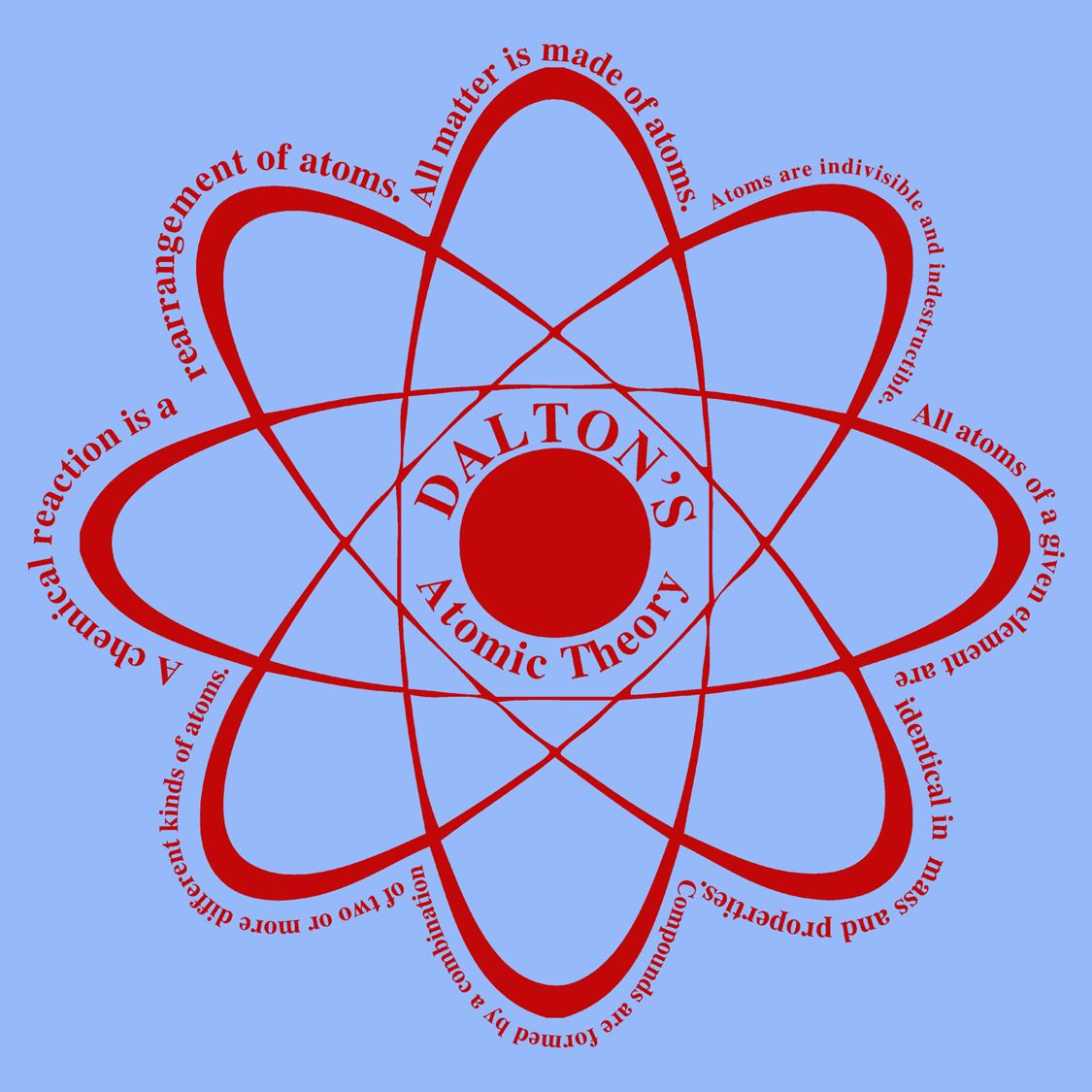
Atoms of an element are identical in mass, size and many other chemical or physical properties, but atoms of two-different elements differ in mass, size, and many other chemical or physical properties.The formation of new products (compounds) results from the rearrangement of existing atoms (reactants).This implies that during chemical reactions, no atoms are created nor destroyed. Atoms are neither created nor destroyed.Atoms of different elements combine in fixed ratios to form compounds.This can also be stated as all the atoms of an element have identical mass while the atoms of different elements have different masses. The properties of all the atoms of a given element are the same including mass.The matter is made up of indivisible particles known as atoms.Learn Dalton’s Atomic Theory from this Video Lecture 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
